

THE SECRETS OF SULFATES IN SKINCARE
Personal care products have become a multi-billion-pound industry in just the UK alone. We’ve spent a staggering one and a half billion pounds on shampoos, conditioners, bath and shower products in the UK last year. With eczema and dermatitis cases rising, what is the real cost of using Sulfates to clean our skin?
What are Sulfates and what are they doing in personal-care products?
Sodium Lauryl Sulfate (SLS), Sodium Laureth Sulfate (SLES) and Ammonium Lauryl Sulfate (ALS), the three most common Sulfates used in personal care products, are a class of chemicals known as detergents which are widely used in shower gels, shave gels, mouthwashes, soaps, shampoo’s and even toothpastes. These chemicals looks like this:
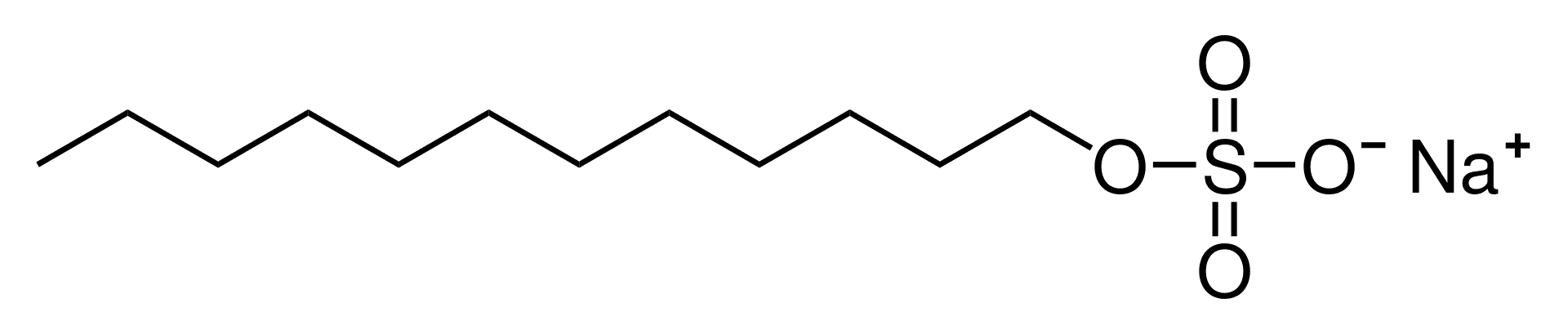
Sodium Lauryl Sulfate (SLS)
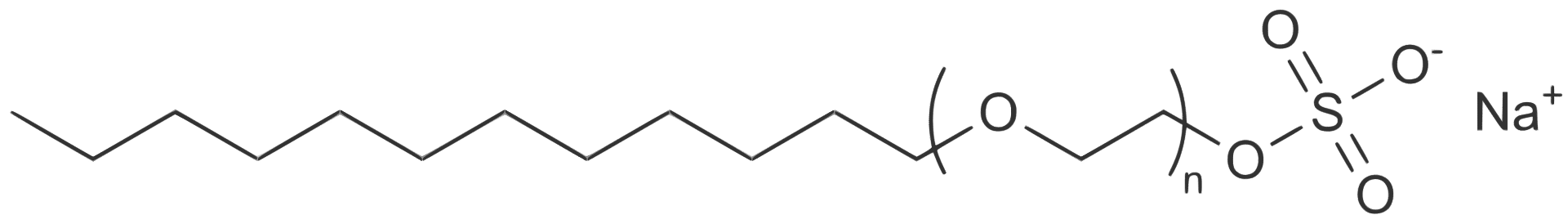
Sodium Laureth Sulfate (SLES)
Ammonium Lauryl Sulfate (ALS)

As with most detergents, these Sulfates have an oil-soluble “tail” (seen on the left) and a water-soluble “head” (seen on the right). This gives it a double purpose, it equally loves water and oil which is its main use in personal care products.
When used with copious amounts of water, the Sulfates are fantastic at helping to separate oils from hair and skin – a little too good sadly. Those oils the Sulfates are trying to remove form an important part of your skin barrier, to keep things out and keep the water in.
So what’s the problem with them?
Chemicals by nature are not intelligent, so the detergents cannot distinguish between the unwanted oil we are trying to remove in order to cleanse ourselves and the necessary oils we need to maintain a healthy skin barrier.
To compound the issue, SLS itself is a comparatively simple molecule and therefore quite small in size, giving it the ability to penetrate straight through layers of skin with ease. When SLS penetrates to the deeper layers of the skin, it comes into contact with the delicate cells that are in the process of being formed into the dermis, causing more damage.
What gives SLS its excellent cleaning ability is also it’s biggest concern –
It’s simply too good at its job and is a well-known irritant.
5% SLS in a product is usually enough to cause irritation in most people, but many people are sensitive to far less. SLS is so well-known at being an irritant, it’s commonly used in lab testing to intentionally harm skin: Following application, scientists can test the efficacy of products intended to heal skin.
Although SLS is a potent irritant, there is no scientific evidence to even suggest that it causes cancer, as it’s wrongly claimed by many. (So that’s something positive… I guess?)
But are they dangerous for my health?
Widespread concern over the past few years into the well-documented irritation by SLS, compelled many companies to look for a gentler detergent with the same properties of SLS, with SLES being created in the process.
Sodium Laureth Sulfate, as well as any other ingredient with an “–eth” in the name, is manufactured through a process called ethoxylation. Unfortunately, in this process of reacting Ethylene Oxide with the starter material, there is an unwanted side reaction which creates 1,4-Dioxane, contaminating not only the ingredient but ultimately the final product. This contaminant can be reduced from the ingredient however as is evident by a study conducted by the EWG, found this contaminant in 46% of products tested.
As for skin and eye irritation, whilst SLS is a slightly more aggressive detergent and will tend to clean better, SLES being the milder form isn’t as irritating but it doesn’t clean as well also.
1,4 Dioxane

The European Scientific Committee on Consumer Safety & U.S. National Toxicology Program has concluded that 1,4-dioxane is ‘reasonably anticipated to be a human carcinogen’ based on numerous animal studies. IARC classifies 1,4-dioxane as ‘possibly carcinogenic to humans’, and the U.S. Environmental Protection Agency (EPA) considers 1,4-dioxane a probable human carcinogen. Exposure to this impurity are linked to tumours of the liver, gallbladder, nasal cavity, lung, skin, and breast. Presence of 1,4-dioxane in cosmetics is of special concern, since it can be absorbed through the skin in toxic amounts.
What about the other Sulfates?
When it comes to the two most common options, though, it doesn’t have to be one or the other. There are other Sulfates out there, such as Ammonium Lauryl Sulfate, the third commonly used detergent in personal care products. It is a very similar chemical to SLS and by all intents and purposes, it is. Along with the inherent disadvantages of being closely related to SLS – the potent irritancy level.
Sulfates by nature do not just disrupt the incredibly complex & delicate skin barrier by removing the oils vital for healthy operation of the skin barrier but also through another more complex and less understood process.
This story continues on Page 2




Leave A Comment