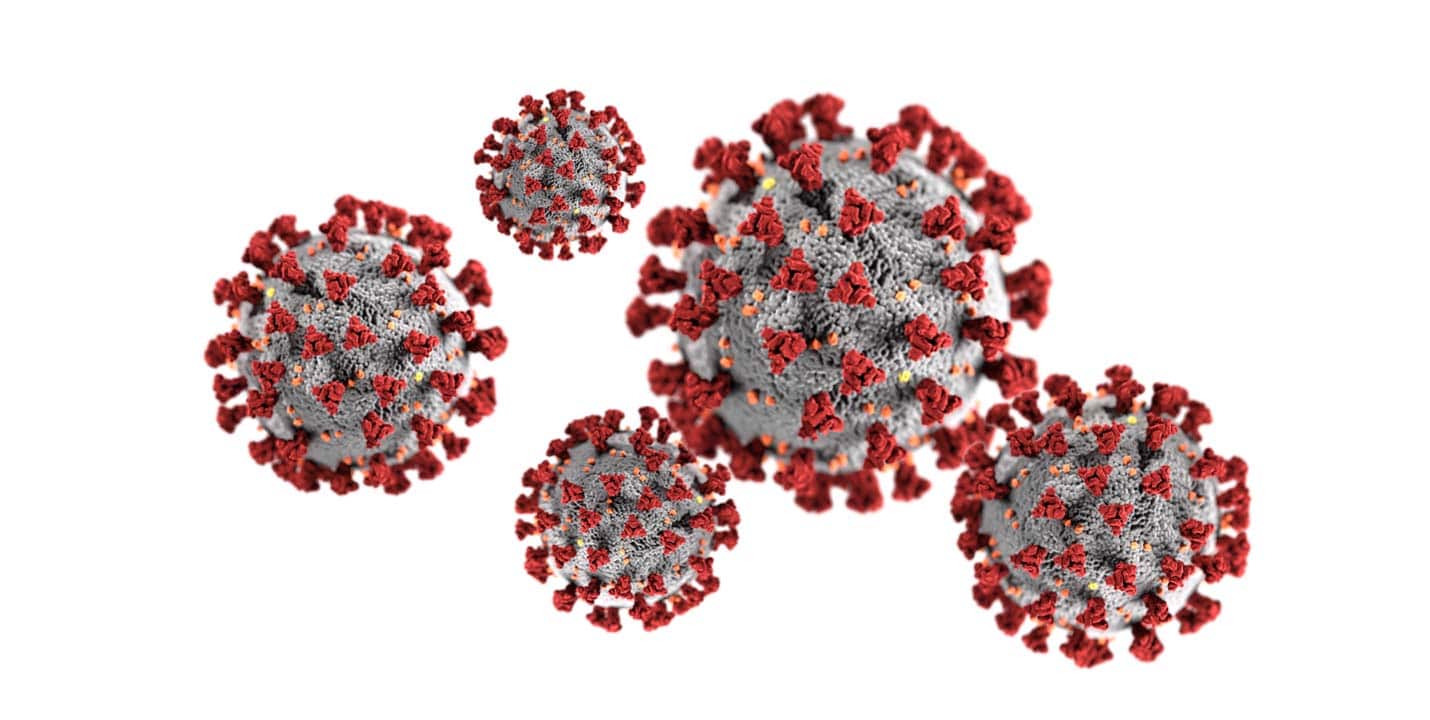

WHY ONE OF THE BEST DEFENCES AGAINST CORONAVIRUS – COVID 19…
IS WASHING YOUR HANDS
As cases of the novel coronavirus have surged, stoking fear and apprehension into the minds of the public, the advice from public health officials is clear: Washing your hands is one of the best defences. Now here is the science behind why this ancient, cheap and effective way to combat the pandemic works.
Respiratory viruses — like the novel coronavirus, the flu, and the common cold spreads easily by droplets from breathing, coughing and sneezing. As our hands touch many surfaces, they can pick up microbes, including viruses. Then by touching contaminated hands to your eyes, nose, or mouth, the pathogens can infect the body through the mucus membranes of your eyes, nose and mouth.
Prevention becomes essential to stopping the spread of the virus because there is no vaccine to prevent it and no anti-virals to treat it. How can such a simple, low-tech solution make a difference?
That’s because when you wash your hands with water and soap – all kinds of it – liquid, solid, bars and even our face wash or shower gel, the ones just for hands… no matter the form, you aren’t just wiping the viruses off your hands and sending them down the drain. You are, in effect, annihilating the viruses.
The Chemistry Behind It
Let’s get this science lesson going then. When developing our 100% natural skincare based on our LiquiCrystal Technology – an emulsion system based upon mimicking the lipid bi-layers in our skin, I became fascinated with the lipid bi-layers found in other systems in nature – like the one found in coronaviruses.
Coronaviruses consist of three key building blocks: RNA, proteins and lipids. The RNA is the viral genetic material — it is like DNA. The proteins have several roles, including breaking into the target cell, assisting with virus replication and basically being a key building block (like a brick in a house) in the virus structure.
The lipids then form a coat around the virus, both for protection and to assist with its spread and cellular invasion. Luckily for us though, there are no strong covalent bonds holding together these key building blocks. These viruses are held together by weak, non-covalent bonds and all it takes is a little crowbar to break these building blocks apart…. Surfactants!
What Are Surfactants?
Surfactants are surface active molecules that have a dual nature. One end of the molecule is attracted to fats and is repelled by water. The other side of the molecule is attracted to water and repelled by fats and proteins. This class of chemicals form the basis of bar soaps and even the coco glucoside & decyl glucoside found in our Active-Performance Face Wash & Deep Cleansing Body Wash.
So How Do Surfactants Kill The Coronavirus?
One side of the surfactant (the one that’s attracted to fat and repelled by water) buries its way into the virus’s fat and protein shell, acting like a miniature crowbar. Fortunately, the chemical bonds holding the virus together aren’t very strong, so this intrusion is enough to break the virus’s coat, pulling the virus apart.
Then the harmless shards of virus get flushed down the drain. The trick is this all takes a little time to happen, and that’s why you need to take at least 20 seconds to wash your hands. The skin is rough and wrinkly, which is why you need a fair amount of rubbing and soaking to ensure the soap reaches every nook and cranny on the skin surface that could be hiding active viruses.
Surfactants don’t just help destroy the viruses – surfactants also help to wash the viruses off your skin too. Washing with plain water might just work, but water “only” competes with the strong “glue-like” interactions between the skin and virus via hydrogen bonds. The virus is sticky and may not budge.
Surfactants outcompetes the interactions between the virus and the skin surface. Soon the virus gets detached and falls apart like a house of cards due to the combined action of the soap and water. Just like that, the virus is gone!
The bottom line:
To sum up, COVID-19, like other coronaviruses are almost like grease-nanoparticles. They can stay active for many hours on surfaces and then get picked up by touch. Then they get to our face and infect us because most of us touch our face frequently.
Water is not effective alone in washing the virus off our hands. Alcohol-based products work better. But nothing beats good old surfactants.
That is why, as I discussed in my previous articles, I hold the humble soap as one of man’s greatest creation. It is, the most basic cleaning agent for humans. What makes it truly the greatest creation of mankind? Surfactants are the most basic weapon in the war to protect our health and to fight disease-causing microbes.
Footnote –
Talking about soaps, I couldn’t pass up the chance to talk about “beauty cream bars” or “beauty cubes” or whatever random term manufacturers like to call these bars of coagulated grease with some synthetic perfume and colour thrown in.
People buy these bars because these bars look like soap. This is not an accident: beauty bars are made to look like soap, for the same reason the manufacturers are very careful not to not to use the word soap on it. All in case it might lead a reasonable person to wield this soap-sized, soap-shaped, soap-smelling thing for the soap-like purpose of washing so that soap shoppers will buy them.
Soap is a surfactant. That miracle of chemistry that lowers the surface tension between your skin and the layer of oily, grimy residue accumulating on top of it, which when applied to your skin and washed off with water, removes the dirt and grime.
By contrast, these lumps of perfumed lard is not a surfactant. It will not kill the coronavirus. Use our 100% natural shower gels, use a decent soap, a purpose made hand wash…. use anything but this!
The science in skincare is our ongoing series helping consumers better understand the science in skincare. We translate the science into a format that is much easier to read, bust the myths and give you a clear, transparent and honest assessment so you can make an informed choice of what goes onto your skin.
Be the first to hear about our new articles by signing up to our email newsletters or by following us on Facebook or Twitter




Leave A Comment